Comirnaty (Pfizer-BioNTech) Vaccine
About the Vaccine
Comirnaty (Pfizer Australia Pty Ltd) is a for-profit vaccine that can prevent people from becoming ill from COVID-19. It does not contain and cannot give you COVID-19. It is an example of an mRNA vaccine, which is a new type of vaccine to protect against infectious diseases. They contain the genetic code for an important part of the SARS-CoV-2 virus called the ‘spike protein’. After getting the vaccine, your body makes copies of the spike protein. Your immune system will then learn to recognise and fight against the SARS-CoV-2 virus, which causes COVID-19. The genetic code is then broken down quickly by the body. Your body will start to build T-lymphocytes and B-lymphocytes (both types of white blood cell that builds antibodies) that will remember how to fight the virus that causes COVID-19 if you are infected in the future.
For more information on mRNA vaccines, visit https://www.cdc.gov/coronavirus/2019-ncov/vaccines/different-vaccines/mrna.html
Safety and Effectiveness of the Pfizer-BioNTech Vaccine
A study published in December of last year tested the safety and efficacy of the Pfizer-BioNTech vaccine on 43,548 participants (21,720 receiving the vaccine and 21,728 receiving a placebo). The study was funded by Pfizer and BioNTech.
This research was an ongoing multinational, placebo-controlled, observer-blinded, pivotal efficacy trial where the researchers randomly assigned persons 16 years of age or older in a 1:1 ratio to receive two doses, 21 days apart, of either the vaccine or the placebo.
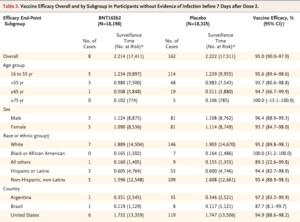
The results of the research were that a two-dose regimen of Pfizer-BioNTech conferred 95% protection against Covid-19 in persons 16 years of age or older. Safety over a median of 2 months was similar to that of other viral vaccines.
Source: Polack et al. (2020) ‘Safety and Efficacy of the BNT162b2 mRNA Covid-19 Vaccine’ The New England Journal of Medicine, accessible at: https://www.nejm.org/doi/full/10.1056/nejmoa2034577
Copyright © 2020 Massachusetts Medical Society.
A study published in February of this year tested the Pfizer-BioNTech vaccination across 99 different centres in the United States and used a sample of 30,420 participants. The study was funded by the Biomedical Advanced Research and Development Authority and the National Institute of Allergy and Infectious Diseases.
This study was a three-phased, randomised, observer-blind, placebo-controlled trial. Individuals at high risk of infection of COVID-19 were randomly assigned in a 1:1 ratio to receive two intra-muscular injections of the Pfizer-BioNTech vaccines or the placebo (15,210 participants in each group) 28 days apart.

The vaccine showed 94.1% efficacy at preventing Covid-19 illness, including severe disease. Aside from transient local and systemic reactions, no safety concerns were identified.
Source: Baden et al. (2021) ‘Efficacy and Safety of the mRNA-1273 SARS-CoV-2 Vaccine’ The New England Journal of Medicine, accessible at: https://www.nejm.org/doi/full/10.1056/nejmoa2035389
Copyright © 2021 Massachusetts Medical Society.
For more studies on the safety and effectiveness of the Pfizer-BioNTech vaccination, see list below:
• Pilishvili et al. (2021) ‘Interim Estimates of Vaccine Effectiveness of Pfizer-BioNTech and Moderna COVID-19 Vaccines Among Health Care Personnel — 33 U.S. Sites, January–March 2021’ Centres for Disease Control and Prevention, accessible at: https://www.cdc.gov/mmwr/volumes/70/wr/mm7020e2.htm
• Dagan et al. (2021) ‘BNT162b2 mRNA Covid-19 Vaccine in a Nationwide Mass Vaccination Setting’ New England Journal of Medicine, accessible at: https://pubmed.ncbi.nlm.nih.gov/33626250/
• Lumley et al. (2021) ‘An observational cohort study on the incidence of SARS-CoV-2 infection and B.1.1.7 variant infection in healthcare workers by antibody and vaccination status’ medRxiv, accessible at: https://www.medrxiv.org/content/10.1101/2021.03.09.21253218v1.full.pdf+html
• Thompson et al. (2021) ‘Interim Estimates of Vaccine Effectiveness of BNT162b2 and mRNA-1273 COVID-19 Vaccines in Preventing SARS-CoV-2 Infection Among Health Care Personnel, First Responders, and Other Essential and Frontline Workers — Eight U.S. Locations, December 2020–March 2021’ Morbidity and Mortality Weekly Report, accessible at: https://miami.pure.elsevier.com/en/publications/interim-estimates-of-vaccine-effectiveness-of-bnt162b2-and-mrna-1
Who can receive this vaccine
People aged 16 years and older can receive this COVID-19 vaccination.
Certain groups of people are prioritised to receive vaccines first because they are at higher risk of:
• exposure to the COVID-19 virus, for example workers in border or quarantine facilities, a healthcare facility or aged-care facility; or
• severe illness and death from COVID-19, for example are older or have underlying medical conditions.
Who should not receive this vaccine
You should not receive this vaccine if you have had:
• anaphylaxis (a type of severe allergic reaction) to a previous dose of the same COVID-19 vaccine; or
• anaphylaxis after exposure to any component of a COVID-19 vaccine.
Precautions for vaccination
People with certain conditions may need additional precautions such as staying for 30 minutes of observation after having their vaccine or consulting an allergy specialist. Tell your immunisation provider if you have had:
• an allergic reaction to a previous dose of a COVID-19 vaccine or to an ingredient of the vaccine;
• anaphylaxis to other vaccines or to other medicines (your provider can check to ensure there are no common ingredients with the COVID-19 vaccine you are receiving); or
• a mast cell disorder.
If you have a bleeding disorder or you are taking a blood-thinning medication (anticoagulant), tell your immunisation provider. Your immunisation provider can help determine whether it is safe for you to have an intramuscular injection, and help decide the best timing for injection.
For more information from the Department of Health about the Pfizer-BioNTech COVID-19 vaccine, see: https://www.health.gov.au/initiatives-and-programs/covid-19-vaccines/learn-about-covid-19-vaccines/about-the-pfizerbiontech-covid-19-vaccine
© Commonwealth of Australia
Adverse Effects
Pfizer and BioNTech conducted tested for the adverse effects associated with the vaccine during the Phase II and Phase III clinical trials. The study enrolled 44,000 participants for an assessment of the immunisation’s safety, immunogenicity, and efficacy endpoints. The research is ongoingly conducted at selected sites worldwide to ensure the diversity of the enrolled population.
For participants aged between 16-55 years of age:
• Injection site pain recorded an 83% frequency after dose 1 and a frequency of 78% after dose 2.
• Fever recorded a 4% frequency after dose 1 and a 16% frequency after dose 2.
• Fatigue recorded a 47% frequency after dose 1 and a 59% frequency after dose 2.
• Headaches recorded a 42% frequency after dose 1 and a 52% frequency after dose 2.
• Chills recorded a 14% frequency after dose 1 and 35% frequency after dose 2.
• Muscle pain recorded a frequency of 21% after dose 1 and a 37% frequency after dose 2.
• Joint pain recorded an 11% frequency after dose 1 and a 22% frequency after dose 2.
For participants aged over 55:
• Injection site pain recorded an 71% frequency after dose 1 and a frequency of 66% after dose 2.
• Fever recorded a 1% frequency after dose 1 and a 11% frequency after dose 2.
• Fatigue recorded a 23% frequency after dose 1 and a 39% frequency after dose 2.
• Headaches recorded a 25% frequency after dose 1 and a 39% frequency after dose 2.
• Chills recorded a 6% frequency after dose 1 and 23% frequency after dose 2.
• Muscle pain recorded a frequency of 14% after dose 1 and a 28% frequency after dose 2.
• Joint pain recorded an 9% frequency after dose 1 and a 38% frequency after dose 2.
For more information, see Pfizer and BioNTech’s ‘Vaccines and Related Biological Products Advisory Committee Briefing Document’ at: https://www.fda.gov/media/144246/download
Myocarditis and Pericarditis
A causal relationship of myocarditis (inflammation of the heart) and pericarditis (inflammation of the membrane around the heart) to the vaccine has not yet been established but is suspected. The TGA has worked with Pfizer to add a warning statement about these adverse events to the Consumer Medicine and Product Information for Comirnaty. This is in response to rare cases following vaccination in Australia and internationally.
These rare effects on the heart typically occur within 14 days of vaccination, particularly after the second dose of Comirnaty and more often in younger men. While cases are usually transient and resolve following rest, some patients require treatment in hospital.
The TGA continues to monitor myocarditis and pericarditis by analysing adverse event reports, working with international regulators and reviewing the medical literature. To 18 July 2021, the TGA received 66 cases of suspected myocarditis and/or pericarditis. The TGA is evaluating these cases in line with internationally accepted criteria to assess whether or not they are myocarditis and/or pericarditis that is likely to be related to vaccination.
Read the TGA’s weekly safety report here.
© Commonwealth of Australia
